what kpa value of glycine to use when making buffer
Module 3.2: Charge & Buffers
- Page ID
- 24655
learning objective
- Calculate the cyberspace charge on a molecule, at whatever pH.
- Understand why solutions of weak acids resist pH changes.
- Calculate the corporeality of a weak acid and its conjugate base of operations to brand a buffer system.
Effect of pH on Charge of Molecules
Since protonation or deprotonation changes the charge of the ionizable group, the pH of a solution will bear on the overall net charge on a molecule. The overall accuse on a molecule as a function of pH can be calculated by following the following steps:
- Place all ionizable groups on the molecule & their accuse when protonated (\(q_{HA}\)) and deprotonated (\(q_{(A^-)}\)).
- Employ the known \(pK_a\) of each group to determine the fraction protonated (\(f_{HA}\)) and deprotonated (\(f_{H^-}\)) at the required pH.
- Calculate the overall charge by summing the contribution of each grouping using the following formula.
\[q_{\text {Total}}=\sum_{i=ane, n} f_{H A} q_{H A}+f_{A^-} q_{A^-}\nonumber\]
walkthrough
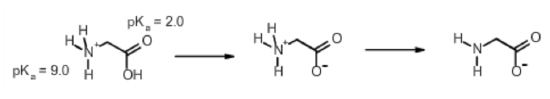
Calculate the net accuse on the amino acid glycine at pH 2.0. Glycine at pH=7.0 is shown beneath:
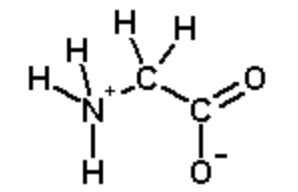
Pace 1. Identify ionizable groups, their \(pK_a\) values, and their charges.
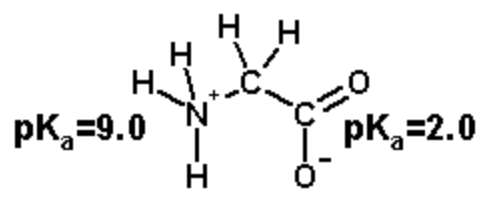
The ionizable groups are the amino and the carboxyl.
The amino group has a \(pK_a\) of 9.0 and the carboxylate has a \(pK_a\) of two.0.
The amino group has a charge of +1 when protonated, 0 when deprotonated.
The carboxylate has a charge of 0 when protonated, -1 when deprotonated.
Step 2. Calculate the fraction protonated (\(f_{HA}\)) and deprotonated (1-\(f_{HA}\)) at the required pH.
These values tin can be obtained quantitatively from the formula: \(f_{HA}\)=i/(one+R), or approximated from a plot of fraction protonated versus pH:
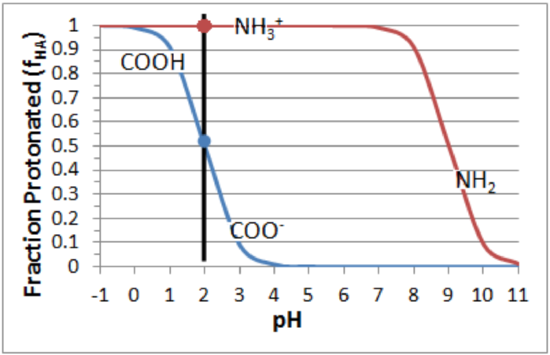
Amino group: \(pK_a\) = nine.0. At pH=2, \(f_{HA}\)=one.0, \(f_{A^-}\)=0.
Carboxyl: \(pK_a\) = 2.0. At pH=2, \(f_{HA}\)=0.5, \(f_{A^-}\)=0.5
Stride 3. Calculate the overall charge by summing the contribution of each grouping: \(q=f_{HA}\times q_{HA}+ f_{A^-}\times q_{A^-}\)
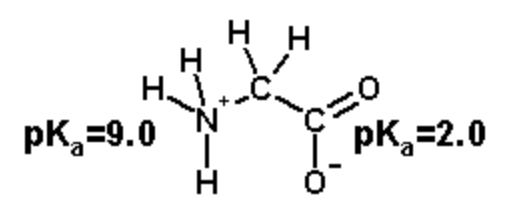
Amino group: \(q_{NH}\)=[1.0x(+1)+0.0x(0)]=+1.0
Carboxylate: \(q_{COOH}\)=[0.5x(0)+0.5x(-1)]=-0.5
Total Charge: q=+1.0 - 0.v = +0.v
Notation that this does non hateful that a molecule of glycine has a charge of +0.5. Instead information technology means that if you lot average over all of the molecules in solution the charge will be +0.5. Based on the above analysis one one-half of the molecules will accept a charge of +1 ( both groups protonated) while one half of the molecules will accept a charge of 0 (amino grouping protonated, acrid deprotonated). The net charge is the weighted average of these ii populations, i.e. 0.5 = 0.5 x (+1) + 0.5 x (0.)
did i get this
1. Select the best approximate the net charge on the amino acid glycine at pH three.0:
a. +one.0
b. +0.i
c. +0.0
d. -0.1
eastward. -ane.0
- hint
-
There are two ionizable groups: the amino group (q=+1 when protonated, q=0 when deprotonated) and the carboxylic acid group (q=0 when protonated, q=-ane when deprotonated).
Estimate the fraction protonated and deprotonated for each group at pH=3.0. For the amino group, \(pK_a\)=9.0, therefore information technology volition be fully protonated at pH=3.0; \(f_{HA}\)=i.0, \(f_{A^-}\)=0. For the carboxylic acrid group, \(pK_a\)=two.0. At pH=iii.0, \(f_{HA}\)~0.i, and \(f_{A^-}|)~0.nine.
The contribution of each grouping to the overall charge is: q = \(q_{HA}f_{HA}+q_{A^-}f_{A^-}\)
- Answer
-
b. (the ratio of products to reactants does non change for a given substance at equilibrium.)
Measuring the \(pK_a\) --Titration Curves
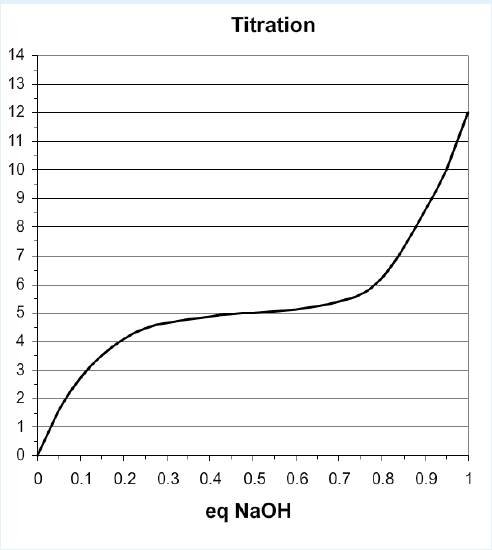
\(K_a\) values, or acidity constants, must be measured by directly experiment, usually with a pH titration. Known amounts of a potent base (NaOH) are added to a solution of weak acid and the pH is measured equally the corporeality of NaOH is added. As the base of operations is added it removes the proton from the acid, too as increasing the pH past removing gratuitous protons from water. A titration curve is shown beneath.
learn by doing
Titration Curve of Monoprotic Acrid
Key features of titration curves:
- Equivalents: the ten-centrality scale for titrations is given in moles of base of operations/moles of acid. Therefore, it varies from 0 to 1 for an acid that releases one proton (monoprotic), from 0 to 2 for a diprotic acid, 0 to 3 for a triprotic acrid.
- Equivalence Point: Consummate deprotonation of the weak acid occurs when the amount of added base is equal to, or equivalent, to the total number of ionizable protons that were originally on the weak acid. This signal in the titration is referred to as the equivalence point. The equivalence point can be used to decide the concentration of the acid.
- Inflection Signal (pH = \(pK_a\)): You can prove from the Henderson-Hasselbalch equation that the smallest change in pH occurs when the pH = \(pK_a\).
walkthrough
Question: The titration of a 0.05 50 solution of phosphoric acid with 1 M NaOH is shown below. Is this a mono, di, or tri-protic acid? What are the individual \(pK_a\)s for each ionization? What is the concentration of the phosphoric acid?
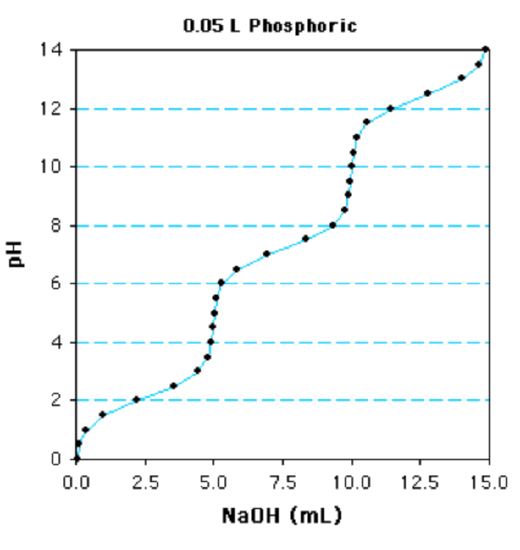
A. Determine the nature of the Acid (monoprotic, diprotic,...).
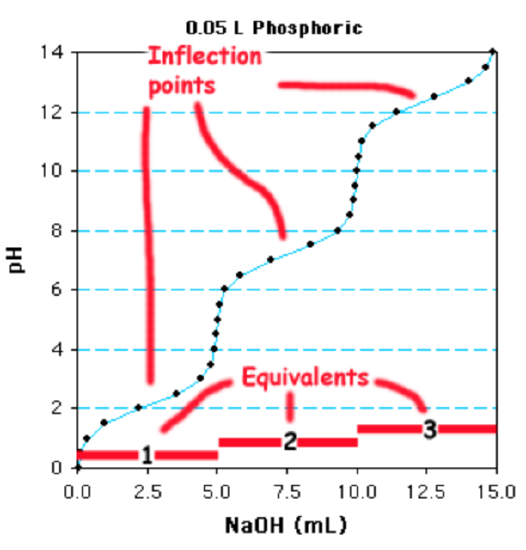
This is a
a. monoprotoic acid considering of one reflection indicate.
b. diprotoic acrid because of two reflection points.
c. triprotoic acid because of three reflection points.
- Answer
-
c. (There are three places on the curve where there is an inflection point.)
In this case there are singled-out inflection points because the \(pK_a\) values are considerably different. In many cases, the individual \(pK_a\) values may exist sufficiently close to each other that multiple inflection points are non clear and a steep change in pH betwixt each ionization is no longer evident. The number of ionizable groups tin can still be easily adamant if one of the ionizations is conspicuously evident. The amount of base required to titrate this group gives the volume of base of operations to titrate 1 equivalent. The full book required, divided past the volume required for one equivalent, volition requite the number of ionizable groups.
Using the phosphate titration as an example, the final ionization begins at 10 ml and is consummate at 15 ml. Therefore five ml is equal to one equivalent. Since 15ml are required for the entire titration, this is a triprotic acid.
B. Determine the \(pK_a\) values.
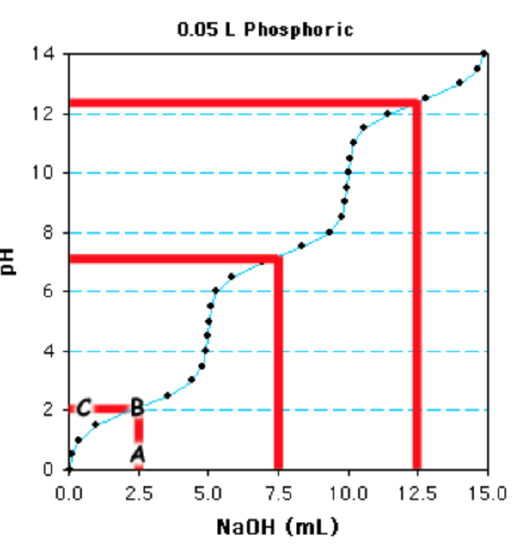
The commencement step is to determine the 1/2-equivalence points - at this point the pH=\(pK_a\) since the concentration of acid equals its conjugate base. This is a triprotic acid and the total book required to titrate the acid is 15ml. Therefore 5 ml (fifteen/three) is 1 equivalence and ii.v ml volition be the get-go 1/ii equivalence bespeak (marked "A" on the graph). The 1/2 equivalence point for the side by side ionization is at vii.5ml (ii.v + 5) and the one/2 equivalence betoken for the final ionization is at 12.5 ml (two.5 + 10).
The adjacent step is to brainstorm from the i/two equivalence point and movement vertically on the plot until the titration bend is met (betoken "B"), then go along horizontally to the y-axis ("C"). This gives the pH at the 1/2 equivalence indicate, ~2.2 in this instance. The other two \(pK_a\) values are obtained in the same fashion, start at 7.5 ml and 12.5 mls, giving \(pK_a\) of ~vii.3 and ~12.iv.
C. Determine the concentration of the acrid.
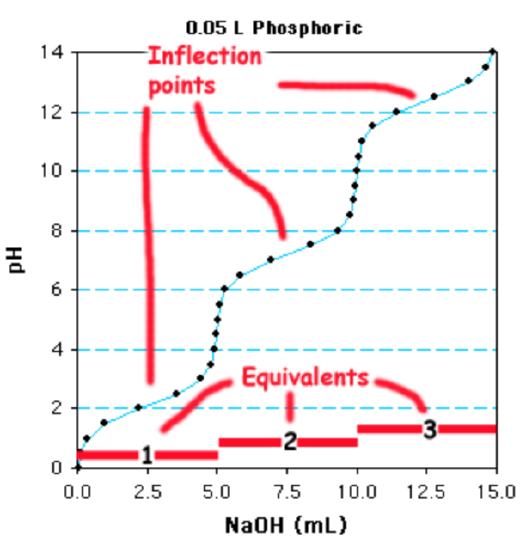
Decide the number of moles of base added: A total of 15ml of 1 M NaOH are required to fully deprotonate the acid. This is a total of 0.015 moles of base (0.015 Fifty x i mole/L = 0.015 moles).
Divide by # of Ionizations to obtain the moles of weak acid: Since this is a triprotic acrid, there are 0.005 moles of the weak acid (0.015/3)
Divide by the volume of the solution to obtain the molarity of the weak acrid. The book of the solution that was titrated was 0.05 L, and so the concentration of the phosphoric acid is 0.005 moles/0.05L = 0.1 M, or 100 mM.
Alternatively, you can recognize that titration of a single grouping of the acrid requires 5 ml of NaOH, which is equivalent to 0.005 moles, carve up by 0.05 L = 0.1 Thousand.
did i get this
i. The following is a titration curve for what blazon of acrid?
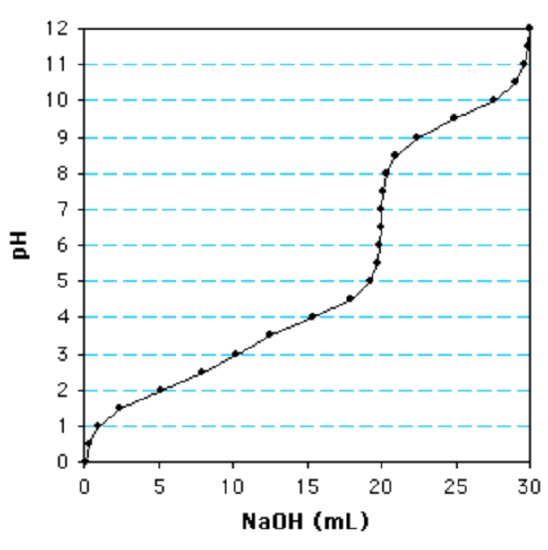
a. Monoprotic
b. Diprotic
c. Triprotic
d. None of these are true
- hint
-
Use the volume required to titrate a group to define the book of one equivalent.
- Respond
-
c. (At that place is more than ane inflection indicate. The last ionization is well separated from the others and shows that ten ml of NaOH are required to titrate one group. Since the total volume of NaOH added is xxx ml, this is a triprotic acid.)
2. Please reply the questions at the bottom of this folio using the following titration bend:
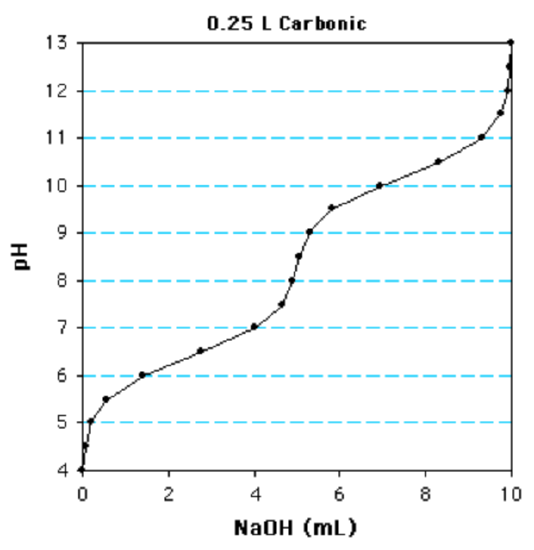
This is a monoprotic/diprotic/triprotic
- hint
-
How many inflection points, or buffer regions, are nowadays?
The first \(pK_a\) value is 6/6.5/7/viii.5/10
- hint
-
The \(pK_a\) is the pH when 1/2 equivalent has been added.
The concentration is 10mM/20mM/40mM
- hint
-
Determine the number of moles of NaOH to titrate one equivalent.
- Answer
-
diprotic (there are two buffer region); half dozen.5 (This is the pH when 1/2 equivalents have been added (2.5 ml)); 20 mM
Buffers
A pH buffer is an acid that resists changes in the solution pH by absorbing or releasing protons. Buffers play an important role in cellular processes because they maintain the pH at an optimal level for biological processes. They are also widely used to control pH in laboratory processes.
The buffering capacity of a solution can exist understood by plotting the fraction of the weak acid that is protonated at unlike pH values. This plot is shown below, plotted with the same orientation as the titration curve, with the pH on the vertical axis. During the titration the fraction protonated (horizontal centrality) will subtract equally base is added; i.eastward. the bend is traced from right to left.
learn by doing
Weak Acids as Buffers
Fraction protonated as a function of pH
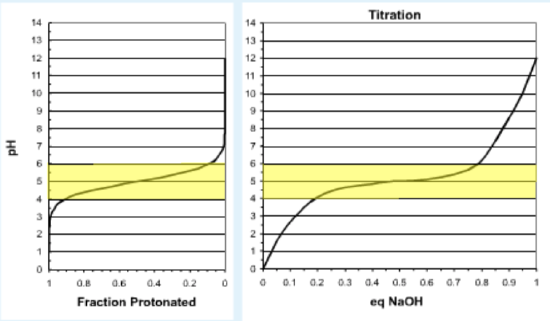
The left effigy shows the fraction protonated versus pH. The correct figure shows a typical titration bend, with the pH as a function of added base. The highlighted area represents the buffer region, which is given by pH=\(pK_a\) +/- 1 unit.
- At the start of the titration the pH is much lower than the \(pK_a\) of the acid, so the acid is fully protonated. Consequently whatever base that is added combines with gratuitous protons and the pH rises sharply without a significant alter in the fraction protonated.
- As more base is added, the pH approaches the \(pK_a\) and the weak acid begins to ionize. The protons that are released from the acid partially neutralize the added base, thus the change in pH as base is added decreases.
- Towards the end of the titration, almost all of the weak acrid is fully ionized and thus any additional base removes protons from the solution, and the pH once more rises sharply.
did i go this
Which region of the curve on the right corresponds to a solution that will not resist pH changes due to the addition of a stiff acid?
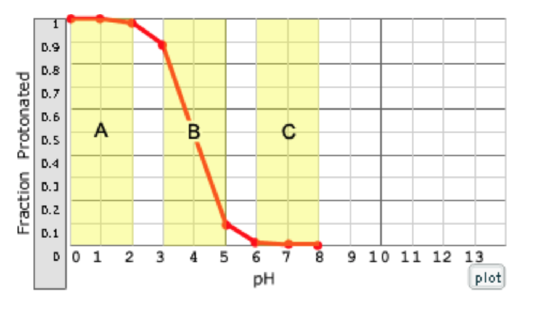
a. A
b. B
c. C
- hint
-
The ability to resist pH changes depends on the ability of the weak acrid to absorb or release protons.
- Reply
-
a. (this region cannot resist changes because the weak acid is fully protonated in region A and can't accept additional protons; in region B, it can resist changes, because both HA and A are present in this region, so the weak acrid can blot boosted protons; in region C, information technology can resist changes because the acid is fully deprotonated in region C and can absorb additional protons.)
In that location are two of import features of buffers:
- Buffering range: This is the pH range over which the pH changes slowly when acid or base is added. The buffer range is considered to be pH values that are one unit below to one unit in a higher place the \(pK_a\). (Hi-lighted in yellow in the effigy in a higher place.)
- Buffer capacity: This is the total amount of base (or acid) that tin exist added to a buffer solution and still have the pH with the buffer range. It depends on the concentration of the weak acid and the starting pH.
Steps in making a buffer
The goal is to brand a solution of a weak acid at a defined pH. Assuming that this pH is inside the buffer region of this acid (pH = \(pK_a\) +/- 1), this solution will resist, or buffer, pH changes. The Henderson-Hasselbalch equation provides some of the necessary data to make a buffer, namely the correct ratio of protonated to deprotonated weak acid to give the desired pH.
\[p H=p K_{a}+\log \frac{\left[A^{-}\right]}{[H A]}\nonumber\]
In gild to obtain a solution of a weak acid at the defined pH, it is necessary to adjust the ratio of [\(A^-\)] to [HA], such that \(pK_a\)+log[\(A^-\)]/HA] equals the desired pH. The required concentrations of [\(A^-\)] and [HA] are defined past the full buffer concentration, [\(A_T\)]:
\[\begin{array}{l}{\left[A^{-}\right]=\left[A_{T}\right] f_{A^-}=\left[A_{T}\correct] \frac{R}{i+R}} \\ {[H A]=\left[A_{T}\right] f_{H A}=\left[A_{T}\correct] \frac{1}{ane+R}}\end{assortment}\nonumber\]
At that place are three means to obtain the indicated ratio of [HA] to [\(A^-\)]:
- Mix the indicated concentration of the acid and its conjugate base to give the desired pH.
- Use [\(A_{T}\)] amount of the acid form of the buffer and add a sufficient amount of a potent base (e.g. NaOH) to generate the required concentration of [\(A^-\)], attaining the desired pH.
- Use [\(A_{T}\)] corporeality of the conjugate base form of the buffer and add a sufficient amount of a strong acid (e.chiliad. HCl) to generate the required concentration of [HA], attaining the desired pH.
In the case of the final two methods, information technology is commonly easier to perform all of the calculations in equivalents and then determine the amount of potent base of operations or stiff acid every bit the number of equivalents ten [\(A_{T}\)]. This approach is especially useful when polyprotic acids are used for buffers.
walkthrough
Example: Make 2L of 0.025 M buffer solution at pH 5.0 using one of the following 2 buffers:
Buffer \(pK_a\) MW
Histidine half dozen.0 155 g/mol
Pyruvate two.5 110 thousand/mol
A. What buffer would you use and why?
Histidine, since its \(pK_a\) is closest to the desired pH of 5.0
B. Determine the amount of the protonated (fHA) and deprotonated (\(f_{A^-}\)) forms that would exist nowadays at pH 5.0.
i. Calculate R kickoff, R=\(10^{(pH-pK_a)}\) = \(10^{-1}\) = 0.1
2. Calculate \(f_{HA}\)= ane/(1+R) = (ane/i.1) = 0.91
iii. Calculate \(f_{A^-}\) = R/(1+R) = (0.one/i.1) = 0.09
Now construct the buffer using i of the iii methods shown on the following pages.
Method A - Mixing of (HA) and (\(A^-\))
\(f_{HA}\)= 0.91
\(f_{A^-}\) = 0.09
The full concentration of all buffer species, [\(A_T\)] is given to be 0.025 moles/L, and the total volume was 2L.
Therefore yous would:
mix 0.91 10 0.025 moles/L x 2 L = 0.0455 moles of HA, or 0.0455 moles x 155 chiliad/mol = 7.05g
mix 0.09 x 0.025 moles/L x 2 L = 0.0045 moles of \(A^-\), or 0.697g
Method B - Addition of Potent Base to HA
\(f_{HA}\)= 0.91
\(f_{A^-}\) = 0.09
Beginning with 1 equivalent (eq.) of (HA) you would need to add together 0.09 eq. of NaOH, which would deprotonate 0.09 eq. of HA to \(A^-\), generating the desired fractions of each species: \(f_{HA}\)= 0.91, \(f_{A^-}\) = 0.09.
The full number of moles of the weak acid is 0.025 moles/L x 2 L = 0.05 moles. Therefore the amount of stiff base to be added is 0.05 moles ten 0.09 = 0.0045 moles. This would be added to 2L of 0.025 1000 (HA).
This process of pH adjustment is illustrated on the post-obit diagram:
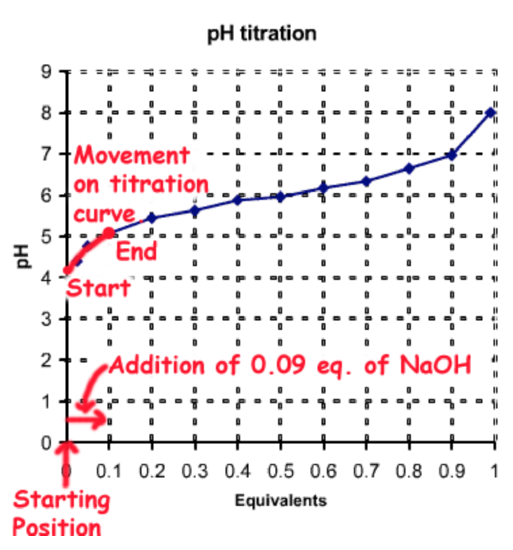
Method C - Addition of Potent Acid to \(A^-\)
\(f_{HA}\)= 0.91
\(f_{A^-}\) = 0.09
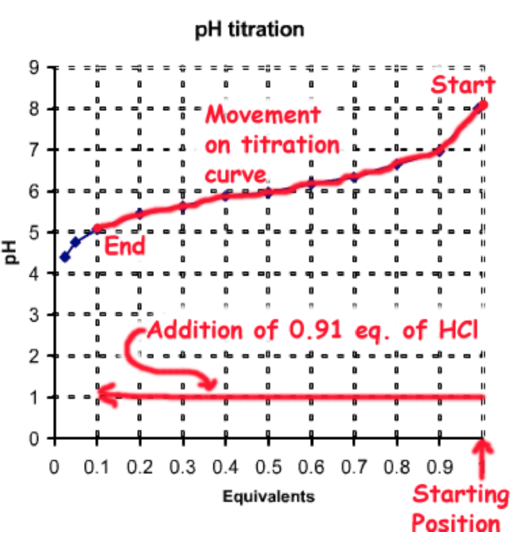
Beginning with 1 equivalent (eq.) of \(A^-\) (e.g. sodium salt) you would demand to add 0.91 eq. of HCl, which would protonate 0.91 eq. of \(A^-\) to HA, generating the desired fractions of each species: \(f_{HA}\)= 0.91, \(f_{A^-}\) = 0.09.
The total number of moles of the weak acid is 0.025 moles/Fifty x ii L = 0.05 moles. Therefore the amount of stiff acid to be added is 0.05 moles x 0.91 = 0.0455 moles. This would be added to 2L of 0.025 Thou NaA.
This procedure of pH adjustment is illustrated on the following diagram:
Polyprotic acids:
- Select an acid that has 1 of its \(pK_a\) values within i pH of the desired pH.
- Summate the fraction protonated and depronated at the desired pH using the selected \(pK_a\)
- Construct the buffer as follows:
- Method A: mix the acid and conjugate base in the ratio defined past the fraction protonated and depronated. The species that you lot would use depends on the \(pK_a\) that was selected. For example, if y'all used a diprotic acid, and used the second \(pK_a\), so yous would mix NaHA (protonated species) and NaiiA (deprotonated species).
- Method B: Addition of strong base. If you lot used the first \(pK_a\) then add \(f_{(A^-\)}\) equivalents of strong base to a solution of the fully protonated weak acid. If y'all used the second \(pK_a\), then add one+\(f_{(A^-\)}\) to the fully protonated acid. The first equivalent converts all of the acid to the singly ionized species, the additional \(f_{(A^-\)}\) generates the right ratio of protonated and deprotonated species.
- Method C: Addition of strong acid. If you used the last \(pK_a\) then add \(f_{HA}\)equivalents of stiff acid to the fully deprotonated weak acid. If you used the next \(pK_a\), and so add together i+ \(f_{HA}\)equivalents of strong acrid, etc.
learn by doing
Buffer Structure With Polyprotic Acids.
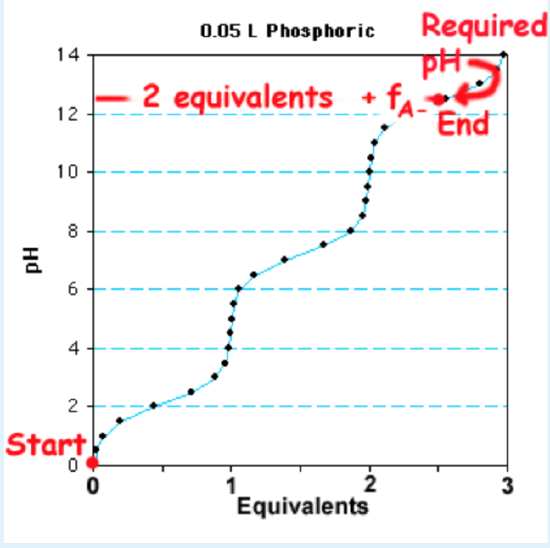
Sample Question: Given 1L of a 0.1 M solution of \(H_3PO_4\), how many moles of NaOH are required to make a buffer with a pH of 12.4? The \(pK_a\) values for phosphate are ii.15, 7.two, 12.iv.
Solution:
ane. The third \(pK_a\) is selected to calculate the fraction protonated and deprotonated since its buffer region includes the desired pH.
2. The Henderson-Hasselbalch is used to calculate \(f_{HA}\)and \(f_{(A^-\)}\), using a pH of 12.4 and the 3rd \(pK_a\). In this example the pH=\(pK_a\), so \(f_{HA}\)= \(f_{(A^-\)}\)=0.5.
iii. The number of equivalents of NaOH to add is two+ \(f_{(A^-\)}\). The get-go ii equivalents convert all of the \(H_3PO_4\) to \(HPO_4^{2-}\), and the remaining \(f_{(A^-\)}\) equivalents establish the right ratio of \(HPO_4^{2-}\) and \(PO_4^{three-}\) to requite the desired pH. This is illustrated on the titration curve shown on the right.
One time the number of equivalents have been determined, the moles of strong base is calculated as:
\[\text { moles }=\text { eq. } \times \left[\mathrm{A}_{\mathrm{T}}\right] \times \mathrm{V}=\left(2+\mathrm{f}_{\mathrm{A}}\right) \times\left[\mathrm{A}_{\mathrm{T}}\correct] \times \mathrm{V}\nonumber\]
where 5 is the total book of the buffer.
REVIEW QUIZ
did i get this
1. A weak acid tin can act as a buffer
a. only at trunk temperatures.
b. at pH values = \(pK_a\) +/- 1.
c. at pH values = \(pK_a\) +/- 2.
d. at pH values = \(pK_a\) +/- 0.1.
due east. at any pH value.
- hint
-
The buffering range is shown in the above diagram.
- Answer
-
B. (This is a useful rule-of-thumb.)
two. In the buffer region
a. added base is neutralized by hydronium ions in the solution.
b. the weak acid is fully protonated.
c. the weak acid is fully deprotonated.
d. added acid protonates the conjugate base of the buffer.
eastward. the ratio of [HA]/[\(A^-\)] is ever constant.
Answer
-
d. (The reason solutions of weak acid are buffered effectually their \(pK_a\) values is because the weak acid can neutralize added base by releasing a proton or neutralize added acid by forming HA; Although the buffer region is characterized by pocket-size change in pH, the ration of [HA]/[\(A^-\)] does change.)
iii. To make a 1M buffer that has a pH that is 1 pH unit of measurement below the \(pK_a\) of the acid, y'all would:
a. add equal amounts of the acid and its cohabit base.
b. add 10 fold higher amounts of the acid than the conjugate base of operations.
c. add x fold lower amounts of the acid than the conjugate base of operations.
- hint
-
Estimate the ratio of HA to \(A^-\) at the desired pH.
- Answer
-
b. (Since the solution is one pH unit more acidic than the pKa of the acrid, you lot would expect the ratio of [HA]:[\(A^-\)] is equal to x:1.)
Source: https://bio.libretexts.org/?title=Under_Construction%2FOLI%2FBiochemistry%2FUnit_2%3A_Biochemistry%2FModule_3%3A_Acids_and_Buffers%2FModule_3.2%3A_Charge_%26_Buffers
0 Response to "what kpa value of glycine to use when making buffer"
Post a Comment